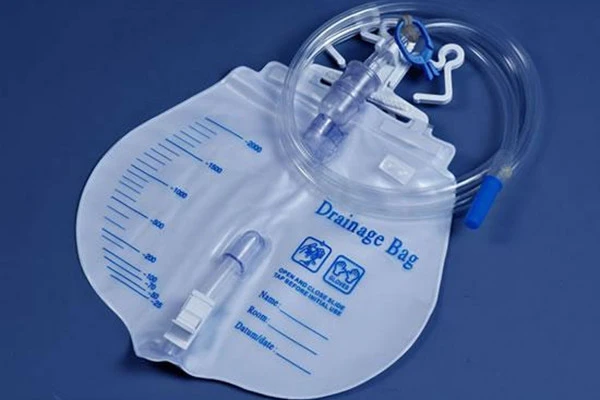
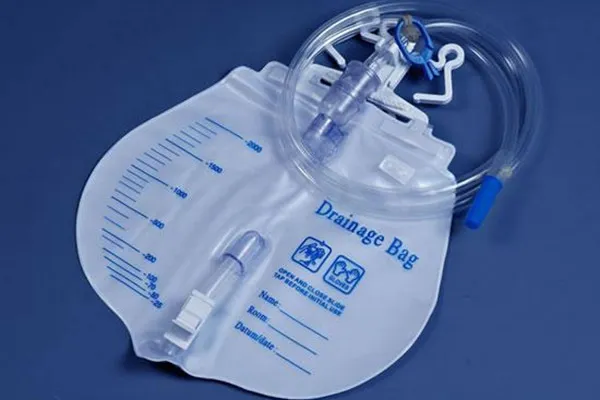
A tiny part that stops big messes: the Drainage Bag Vent
If you work with medical or industrial drain bags, you already know the paradox: you need air to escape fast, but not a single drop of liquid should. That’s the everyday job of a hydrophobic vent plug. I’ve toured a few cleanrooms over the years, and—surprisingly—this small, press-fit component often drives the success rate of entire drainage systems.
Origin: Rm. C-1301, Hyde Park Plaza, No. 66 Yuhua W. Road, Shijiazhuang, 050056 China
What it is and why it matters
The Drainage Bag Vent is a hydrophobic, precisely press-fit plug that equalizes pressure as a bag fills, preventing collapse, burping, or—worst—liquid leakage. Many customers say it’s “install-and-forget.” In fact, good vents cut operator intervention during draining procedures, and make bag swaps faster and cleaner.
Industry trends (quick take)
- Shift to sintered hydrophobic polymers (PP, PE, ePTFE) with tighter pore control and higher Water Entry Pressure (WEP).
- Cleanroom assembly under ISO 13485 systems for medical supply chains.
- Customization: color-coding, snap-fit geometries, carbon layers for odor management (wound/urine applications).
- Sustainability pressure: lighter plugs, recyclable carriers, fewer parts.
Technical specifications (typical)
| Material | Hydrophobic PP/PE (sintered), optional ePTFE laminate | Biocompatibility per ISO 10993 (real-world use may vary by grade) |
| Nominal pore size | ≈ 0.2–20 µm | Selected per flow and retention needs |
| Airflow @ ΔP | ≈ 0.5–5 L/min @ 70 mbar | Geometry-dependent |
| WEP (water entry pressure) | ≈ 0.8–2.5 bar | Higher = better liquid holdoff |
| Operating temp | -20 to 90 °C (PP/PE) | Process-dependent |
| Sterilization | ETO, Gamma; Steam case-by-case | Verify per design |
How it’s made (short process flow)
Materials: medical-grade PP/PE powders; optional ePTFE membrane. Methods: controlled sintering for porosity, precision turning/molding of carriers, press-fit assembly to exact tolerances. Testing: pore size per ASTM F316, airflow calibration, WEP verification, bacterial retention (ASTM F838), 100% visual and random leak testing. Service life: typically single-use; shelf life ≈ 3–5 years in dry packaging.

Where it’s used
- Medical: urine and wound drainage bags, suction canisters, ostomy pouches.
- Laboratory/bioprocess: carboys, waste bags, sample containers.
- Industrial: paint/ink and chemical waste bags, detergent reservoirs, appliances.
Advantages you can feel on the floor
- No-drip hydrophobic barrier; stable pressure equalization.
- Press-fit simplicity—fast line changeovers.
- Customization: barb/nozzle IDs, cap colors, odor-adsorbing layers.
- Cleanroom-ready production; traceability for audits.
Vendor comparison (condensed)
| Criteria | Chinaporousfilters | Generic Importer | Local Mold Shop |
| Porosity control | Tight (ASTM F316 data) | Varies | Limited test data |
| Biocompatibility docs | ISO 10993 material options | Sometimes | Rare |
| Customization speed | Fast, iterative | Slow | Fast for tooling, not porous media |
| Cert readiness | ISO 13485/9001 frameworks | Varies | Limited |
Real-world note and test data
A European ostomy-bag maker told me they saw ≈42% fewer leak returns after switching to a Drainage Bag Vent with higher WEP (≈2.0 bar) and an activated-carbon cap. Internal QA snapshots (n=30 lots) showed airflow consistency within ±8% and ASTM F838 retention of >99.99% for Brevundimonas diminuta. To be honest, your mileage may vary—but the direction of travel is clear.
Customization checklist
- Geometry: OD/ID for press-fit chambers, snap or thread adapters.
- Porosity: target pore size and WEP for your fluid.
- Add-ons: odor control, tamper seals, color coding, sterile packaging.
- Docs: COC, lot traceability, material biocompatibility summaries.
Certifications & standards: ISO 13485/9001 quality systems; materials tested to ISO 10993 and USP Class VI; cleanroom assembly per ISO 14644; performance characterization referencing ASTM F316 and F838. Regulatory submissions remain the device owner’s responsibility.
Authoritative citations
- ISO 13485:2016 – Medical devices QMS.
- ISO 10993 series – Biological evaluation of medical devices.
- ASTM F316 – Pore size characteristics of porous materials.
- ASTM F838 – Bacterial retention for membrane/porous filters.
- ISO 14644 – Cleanrooms and associated controlled environments.
Post Time: Oct . 21, 2025 16:20